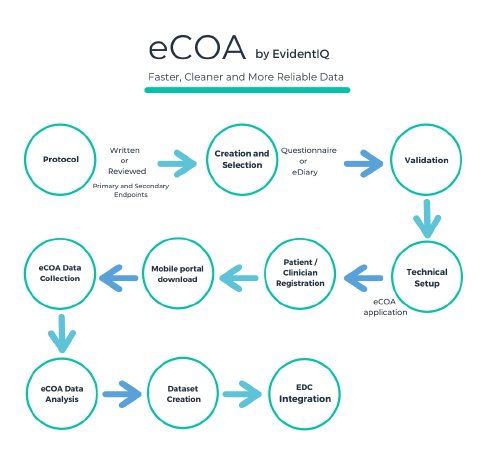
With its comprehensive eCOA solution, the new data science group, which has only recently been built by merging long-established providers XClinical, Carenity and Fortress Medical, has set out to fuse data science services and a scalable software platform. EvidentIQ customers benefit from an end-to-end eCOA package allowing to collect any patient / clinician / observer reported outcome data in a streamlined, compliant and efficient way. The solution can be used stand-alone or fully integrated into existing eClinical technologies, such as EDC systems. The EvidentIQ eCOA offering supports a bring-your-own-device (BYOD) policy and works on any iOS/Android phone.
Combining a software platform with scientific services
EvidentIQ’s combined offering of a platform with a scientific service offering attached to it empowers LifeScience professionals to conduct virtual and hybrid clinical trials. Unique eFeasibility solutions help optimize clinical trials and accelerate patient recruitment with a patient-centric approach.
EvidentIQ relies on Data Science and robust technologies to custom the design of clinical trials optimizing Product Profiles, Endpoints, Protocols, ICFs, and eCRFs. The package facilitates a complete digitalized patient enrollment journey for clinical trials from Multi-channel Pre-screening over eConsent Management to the Clinical Data Capture in the EDC.
Steady Market Growth for eCOA
Andreas Weber, CEO of EvidentIQ, points out the substantial market growth for Electronic Clinical Outcome Assessment:
“Most market analyses expect a CAGR of more than 15% for the eCOA market until 2027, and a global market volume of approximately 2.5 billion USD by then.”
When asked for the main drivers causing this growth Weber sees multiple developments besides the global Covid-19 pandemic.
“Keep in mind there is a high demand for clinical trials in emerging markets and consistently high R&D spendings in the pharmaceutical industry. Besides FDA and EMA are taking a tougher stance on COA data collection meeting regulatory quality guidelines.”
“Our new eCOA offering demonstrates perfectly how we create value for our customers and in doing so build a unique market position for our group. We empower life science companies of all sizes to transition from outdated, error-prone paper-based processes to digital data capture and data processing.”